The iron that contains less than 2% of carbon is called steel whereas which contain more than 2% of carbon is known as pig iron. When iron ore is processed with coke in a blast furnace, pig iron is obtained.
process used to separate iron from iron ore
Copper processing is a complicated process that begins with mining of the ore (less than 1% copper) and ends with sheets of % pure copper called cathodes, which will ultimately be .
The main plants involved in such a route hare been treated and the main process steps have been described: sintering of the iron ores, coal treatment to obtain the coke, pig iron production by the ...
Metal Extraction Concepts Key Concepts. ... The more stable the compound is, the more energy is required to separate the metal in the metal ore. ... Calcium occurs before iron in the activity series so calcium is the more active metal. Infer the relative stability of the compounds each metal will form.
Metal ores are formed in the process of ore genesis, and they are extracted through mining. Extractive metallurgy is the practice of removing valuable metals from an ore and refining the extracted raw metals into a purer form. Hydrometallurgy uses aqueous solutions to extract metals from ores ( leaching ).
It's a major ingredient in steel. Here's what the USGS has to say about iron ore: Iron ore is the source of primary iron for the world's iron and steel industries. It is therefore essential for the production of steel, which in turn is essential to maintain a strong industrial base. Almost all (98%) iron ore is used in steelmaking. Iron ore is mined in about 50 countries.
Iron ore is used to make iron and steel and iron is produced in a blast furnace by reducing iron oxides with carbon and it is the carbon that removes the oxygen from the iron oxides – the carbon is known as the reducing agent.
2. What is iron ore used for? Iron ore is usually smelted to produce pig iron (metallic iron), which is used to make steel. It is for this reason iron is perhaps one of the most important metals used in developed and developing is also wrought iron (low in .
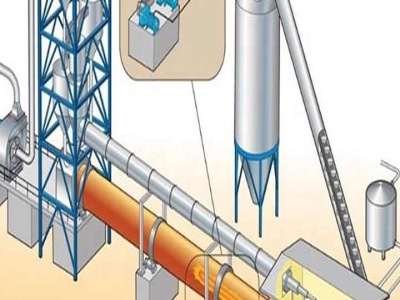
Oct 10, 2017· Source: The German Steel Federation (WV Stahl) In order to make steel, iron must be extracted or won from iron ore. Blast furnacing, smelting and direct iron reduction are the current ironmaking processes. Ironmaking Blast Furnace. Iron is made by reacting iron ore (iron oxide and impurities), coke (a reductant) and limestone (CaCO 3) in a blast furnace.
In essence, all primary iron and steel production processes are based on the same chemical reactions. In iron ore, iron is presents in its oxidised form. In iron reduction processes, a reducing agent, based on carbon or hydrogen, removes the oxygen from the iron.
Iron ore Wikipedia. Iron ores are rocks and minerals from which metallic iron can be economically extracted. ... Iron ore is the raw material used to make pig iron, which is one of the main raw materials to make steel—98% .... rock, but are considerably cheaper to mine and process as they require less beneficiation due to the higher iron content.
Iron removal system schema. Iron is often found in water in complexed forms. In order to be eliminated, iron complexed requests a coagulation stage, which comes in between oxidation and filtration. Remark : Thanks to microorganisms, it is possible to remove iron from water by biological way.
to the blast furnace to remove the impurities in the iron ore. Calcium carbonate is decomposed by heat in the furnace to give calcium oxide and carbon dioxide. This is called thermal decomposition (see examples of other carbonates). ... and is used for road construction.
Let us try to separate kerosene oil from water using a separating funnel. ... • In the extraction of iron from its ore, the lighter slag is removed from the top by this method to leave the molten iron at the bottom in the furnace. Share on Facebook Share on Twitter Share on Google+.
As a result, this type of iron ore ore must be concentrated before it can be used to produce steel. Magnetite ore's magnetic properties are helpful during this process. Learn to profit from base ...
Sintering is a process used to agglomerate iron ore fines in preparation for . Iron processing Britannica. Iron processing, use of a smelting process to turn the ore into a form from which products can be .. The earliest kind of firing equipment was the shaft furnace. Iron ore mining in Australia: Wirtgen Surface miners are main mining
Posts Related to how to separate chromite fines from lateritre ores conventional metallurgical process of smelting of chromite ores chrome ore smelting and fine crushing machines manufacturer The conventional metallurgical process of smelting of chromite ores in submerged arc furnaces requires the use .
It is formed when iron ore is refined in a furnace in the presence of charcoal, limestone and air. It is the basic form of iron used to make decorative wrought iron items. When the pig iron is further refined and a minimum amount of carbon is added, the crystalline structure of .
Iron, Steel, and Aluminium. The process of slowly heating steel and holding it at a particular temperature to modify crystal structure, and then cooling it. Rate of heating and cooling and the temp raised to depend on the type of steel and the desired properties.
The technology required to separate iron from its ores and convert it into durable and useful objects is far more complicated than that needed to work successfully with copper and bronze. For one thing, it requires a temperature of about 3650 degrees Fahrenheit (about 2020 degrees Celsius) to cause iron to melt sufficiently so that it will flow.